A drug that was originally developed to treat diabetes and severe overweight might also help people with nicotine dependence, concludes new research from the University of Copenhagen.
Smoking is one of the greatest threats to public health. According to new data from the Danish Health Authority, 15,920 Danes die every year because of their addiction to cigarettes.
And 75% of smokers want to quit.
So why don’t they? The answer is: nicotine. It is one of the most addictive known substances.
According to the Danish Health Authority, almost one in four Danes is addicted to at least one nicotine product. And young people’s use of snuff and electronic cigarettes is increasing.
Unfortunately, there is no treatment for nicotine cravings.
But new research from the University of Copenhagen might be able to change that.
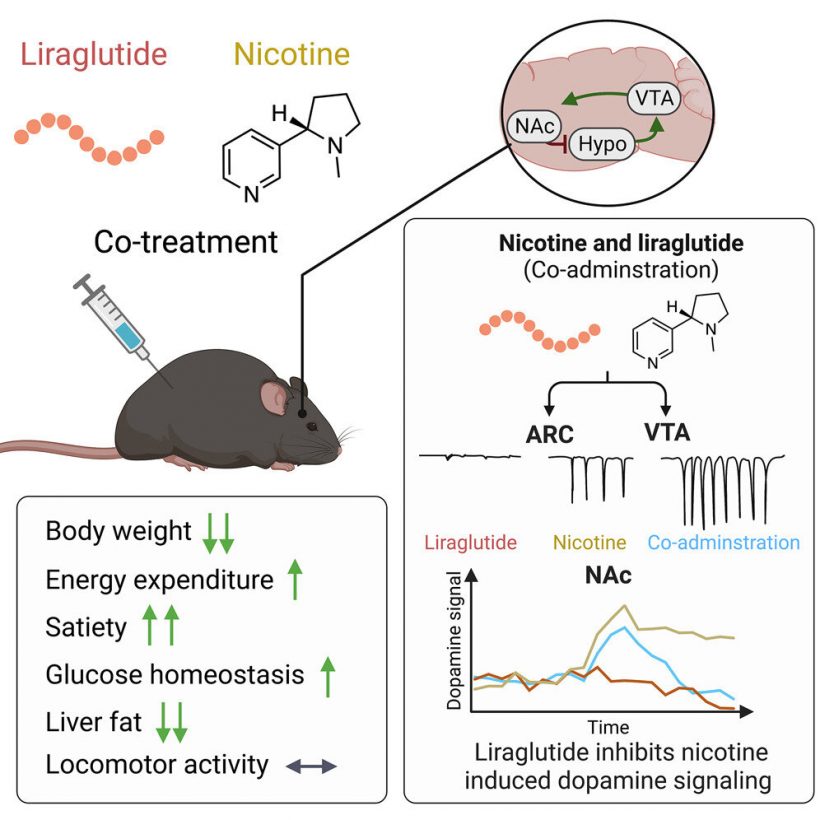
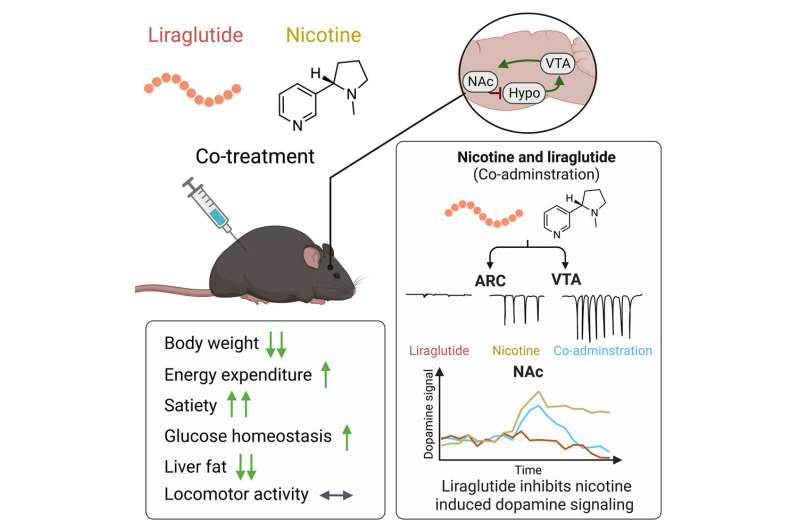
“In connection with a mouse study focusing on overweight, we discovered, more or less by accident, that a well-known drug, liraglutide, used to treat diabetes and overweight, affects the impact of nicotine on the brain. Liraglutide inhibits the effect that nicotine has on the brain’s reward system,” says the main author of the study, Associate Professor Christoffer Clemmensen from the Novo Nordisk Foundation Center for Basic Metabolic Research at the University of Copenhagen.
He explains that GLP-1 and nicotine are the two substances that affect the brain’s reward system. Nicotine from tobacco, for example, releases a large amount of dopamine in the brain, which acts as a kind of reward. This is one of the reasons why it is so hard to quit nicotine products.
“Seeing as our study has a different focus, i.e. to determine whether the two substances interact to promote weight loss, we did not pursue the surprising effect on the dopamine system, and more research is therefore needed to determine whether treatment with liraglutide will cause mice to ingest less nicotine,” adds Christoffer Clemmensen.
New clinical trials focussing on nicotine dependence
In the study, which has been published in the journal Cell Reports, the researchers studied two known appetite inhibitors: GLP-1 and nicotine. GLP-1 is used in various diabetes and weight loss drugs.
The researchers already knew that nicotine increases energy expenditure, and many who quit nicotine products subsequently gain weight.
Together, the two substances appear to promote weight loss, while GLP-1 alone seems to reduce nicotine cravings. And, according to the international database of clinical trials, many researchers are currently testing whether GLP-1-based drugs can help people quit smoking.
Collaboration with UCPH brain researchers
Christoffer Clemmensen explains that it was only recently discovered that a combination of GLP-1 and nicotine has a greater effect on the body than either of the two substances alone.
The two substances affect the same neurons in specific parts of the brain, and the researchers have been able to demonstrate that GLP-1 affects the body’s response to nicotine.
“Initially, we therefore wanted to see whether a combination of nicotine and GLP-1 could effectively reduce overweight. Our animal models showed some interesting effects. So we did more tests focussing on the reward and appetite regulating systems,” says Christoffer Clemmensen.
“We were surprised to learn how extensively GLP-1 and nicotine interact. It is a well-known fact that both substances affect food intake, and this effect seems to be even greater when you combine the two substances. It not only reduces food intake; it also causes the body to release less dopamine, reducing nicotine cravings,” he says.
The trials focussing on the brain’s reward system were conducted together with the Gether Lab at the Department of Neuroscience, University of Copenhagen, who measured dopamine release in the brain.
The researchers are not planning to conduct clinical trials of the interaction between GLP-1-based drugs and nicotine, but they hope their work will encourage pharmaceutical companies to consider the potential of the area.
More information:
Sarah Falk et al, GLP-1 and nicotine combination therapy engages hypothalamic and mesolimbic pathways to reverse obesity, Cell Reports (2023). DOI: 10.1016/j.celrep.2023.112466
Journal information:
Cell Reports
Source: Read Full Article